FLOW CYTOMETRY
CASE STUDY
Background
B cell depletion therapies are critical interventions for autoimmune diseases and hematologic malignancies. For sponsors developing these therapeutics, accurately measuring residual B cells at extremely low concentrations – down to 10-20 cells/μL – is essential for understanding pharmacodynamic effects and determining optimal dosing regimens. However, achieving this level of sensitivity while maintaining statistical precision presents significant technical challenges in flow cytometry.
The challenge intensifies when considering that Flow Cytometry is subject to Poisson counting statistics-rare cells do not distribute evenly.

According to formula to achieve 10% precision (coefficient of variation) on rare cell populations, you need to acquire at least 100 events. However mathematical formula doesn’t factor in natural variability that comes from handling cells, thus overestimating low precision. At concentrations of 10 cells/μL, this means analyzing substantial sample volumes – and every source of variability becomes a potential obstacle.
Example:

Absolute Counting Method
Analysis tube contains pre-determined number of beads (from Manufacturer) that gets mixed in blood. Antibody master mix is incubated with blood and Lysis buffer is added and incubated. Without any wash and cells are then collected on cytometer. Optimisation of the method heavily relies on correct volume of blood, selection of incubation time and temperature and Selection of correct Lysis buffer volume, incubation time and temperature.
The Challenge
A pharmaceutical client needed a validated method to enumerate B cells with:
- Sensitivity down to 10-20 cells/μL
- Targeted Precision of 10% CV
- Extended acquisition time (15-20 minutes analyzing 200μL to 2mL samples)
The fundamental problem: how do you create appropriate controls and validation samples for such rare cell populations?
Standard approaches fell short:
- Commercial controls such as a low BC specify B cell concentrations ≥100 cells/μL – an order of magnitude too high for the required sensitivity
- Diluted blood samples don’t accurately represent the sample matrix after red blood cell lysis, as extreme dilution (20-50 fold) produces far less debris than actual study participant samples would as well dilute haemoglobin
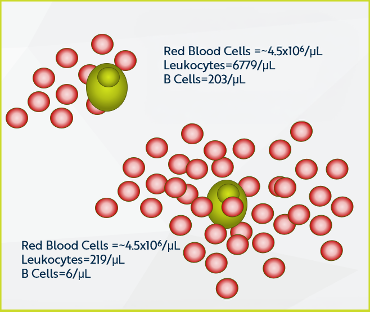
- Multiple sources of variability threatened precision targets:
- Population separation quality
- Blood lysis procedure
- Debris and excess of haemoglobin
- Analyst-to-analyst variation
- Instrument variability and temperature effects
- Sample logistics and handling
The client needed cell concentrations that would naturally contain only 10-20 B cells/μL in a realistic matrix environment – but how do you reliably create and validate such a system?
Spectral Flow Cytometry: From Trade-Offs to Total Clarity?
Resolian combines the Cytek Aurora Full Spectrum Profiling platform with 25+ years of flow cytometry expertise to deliver complete immune clarity and confident, regulatory‑ready decisions for cell & gene therapy programs.
Resolian’s Solution
Resolian’s flow cytometry team developed an innovative approach: creating a depleted whole blood matrix that mimics real-world sample conditions at ultra-low B cell concentrations.
Method Development Strategy:
- Matrix Creation: Rather than simply diluting blood (which changes debris characteristics), Resolian depleted ALL leukocytes from whole blood by 98.2%, reducing the concentration from 4.91×10⁶ cells/mL to 2.2×10⁴ cells/mL. This was confirmed using NC-200 cell counting
- Triple Quantification Methods:
- Volumetric measurements using the instrument’s built-in capabilities
- TrueCount bead standardization for absolute counting
- Nucleocounter NC -200
- Bead and volumetric measurements of instrument was used to confirm this.
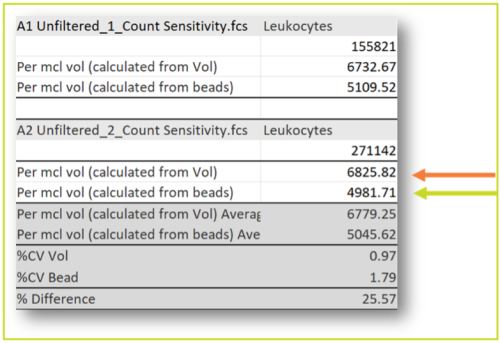
Out of three methods there was a strong agreement between the two- Bead and Nucleocounter method therefore we proceeded with Nucleocounter method in further assessment.
- Spiking Approach: The team spiked varying concentrations of whole blood (100% down to 0.03125%) into this depleted matrix, creating a dilution series spanning from ~150 cells/μL down to 0.5 cells/μL – achieving the target sensitivity of 1 cell per 2μL.
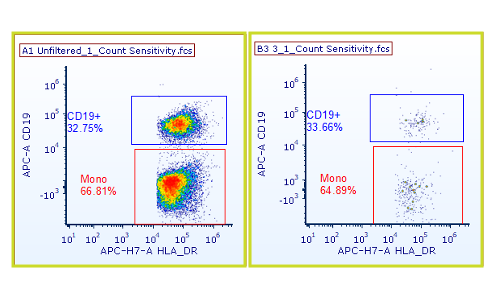
- Validation: The team assessed:
- Linearity across the full concentration range (r² = 0.996)
- Precision at each concentration level
- Recovery efficiency
- Bead-based method was infinitely superior to Volumetric measurement of instrument.
Results
The validated method successfully met all client requirements: Sensitivity: Demonstrated detection down to 0.5 cells/μL (1 cell per 2μL) – well below the 10-20 cells/μL target Linearity: Excellent correlation (r² = 0.996) across the entire concentration range from 0.5 to 150 cells/μL Precision:- Overall average %CV: 5.27% – exceeding the 10% target
- Individual replicate %CVs ranged from 0.54% to 9.12%
- Even at the lowest concentration (0.47-0.48 cells/μL), achieved 2.11% CV
- Average recovery: 97.21%
- Range: 85.78% to 104.26% across concentrations
- Consistent performance from highest to lowest levels
- Bead-based measurements showed good agreement with Nucleocounter
Impact
This validated high-sensitivity B cell counting method enables sponsors to:- Monitor deep B cell depletion in clinical trials with confidence in data quality at ultra-low concentrations
- Make informed dose-escalation decisions based on reliable pharmacodynamic measurements
- Support regulatory submissions with a robust, validated bioanalytical method
- Reduce sample volume requirements while maintaining statistical power
Resolian partners with pharmaceutical and biotech companies to solve complex bioanalytical challenges in flow cytometry, enabling faster, more reliable decisions in drug development.
Contact us to discuss your method development needs.

