Impurity Identification
& Characterization
An interdisciplinary team offering precise and versatile Identification & Characterization services.
Resolian excels in Impurity Identification & Characterization, offering comprehensive solutions regardless of your sample matrix.
An interdisciplinary Impurity Identification & Characterization team collaborates closely with Analytical Chemistry & Trace Analysis, Extractables & Leachables, and Foreign Matter Analysis teams to identify critical degradation products throughout the development cycle.
Incorporating advanced instrumentation, Resolian specializes in the structural elucidation of active components and process impurities during all phases of development lifecycle for both small and large molecules.
These innovative approaches provide online separation and detection of compounds, expanding capabilities to tackle your impurity identification challenges comprehensively.
Reach out to us directly to learn more about Impurity Identification & Characterization services.
Services in Detail
Impurity Identification Regardless of Matrix: Offering you expertise to identify and characterize impurities in diverse matrices, from macroscopic particles to the smallest chemical components.
Visible/Subvisible Particles: Resolian’s Foreign Matter Analysis team excels in handling visible and subvisible particles, ensuring thorough analysis.
Non-Volatile Components: Resolian employs cutting-edge technologies for both small molecules and large biomolecules in highly complex formulations, including UPLC-IM-HRAMS and 2D-UPLC-HRAMS, enabling confident characterization.
Semi-Volatile Components: Resolian utilizes CI and EI GC-MS complementing the identification of semi-volatile components, ensuring a comprehensive impurity profile.
VOCs and VVOCs Identification: Resolian’s HS-GC-MS, equipped with cryogenic capabilities, facilitates the identification of very volatile organic compounds (VVOCs).
Impurity Identification
Characterisation and monitoring of impurities using Ultra-Performance Liquid Chromatography and High Resolution Mass Spectrometry (UPLC-HRMS).
Knowledge of the impurities and degradation profile of drug substance
and excipients is critical in understanding the stability, safety and efficacy of pharmaceuticals and biopharmaceuticals.
To identify, with a significant degree of certainty, low level impurities in the presence of high level components requires the combination of multiple analytical techniques and expert interpretative skills.
Our approach uses advanced chromatography and mass spectrometry platforms combined with innovative powerful software packages.
Together, the team of highly skilled scientists interrogates and interprets the data generated to elucidate the structure of individual or multiple
unidentified components.
CASE EXAMPLE:
CHALLENGES
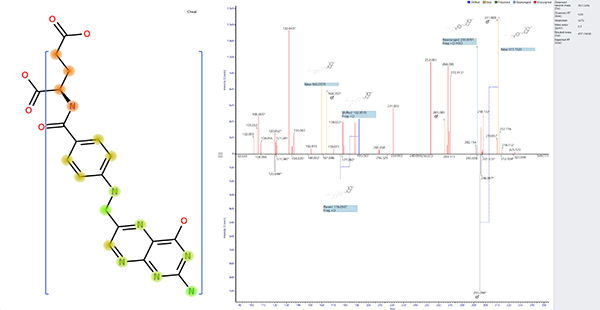
One example of quantifying impurities at very low levels involved an unknown degradation product of a human peptide hormone.
The sponsor had observed a new peak in the standard LC-UV chromatography during the stability assessment of the product.
SCIENCE
Our scientists established the sponsor’s method on to an Acquity UPLC coupled to a Waters SynaptTM G2 mass spectrometer. The method was modified for maximum sensitivity and the data interpretation focussed on specific regions within the chromatographic run.
The data independent acquisition (DIA) enabled the simultaneous identification of components using the high resolution, accurate mass MS/MS fragmentation information (Figures 2 and 3).
OUTCOME
The data identified peptide sequences related to the human peptide hormone.
These peptide sequences identified a degradation pathway for this compound and highlighted a potential stability issue that previously had not been considered.
A VALUE ADDED SERVICE
Following the identification of an unknown species in your product, we can develop and validate appropriate assay and related impurity methods to support release testing and stability studies.
Our structural elucidation capabilities are also used to strengthen our Extractables and Leachables services in identifying unknown compounds from extraction studies.