Analytical Chemistry
& Trace Analysis
Providing a comprehensive range of Analytical Chemistry services for diverse pharmaceutical applications.
From analytical method development and validation to routine testing and trace impurity analysis, Resolian is your trusted partner for ensuring product safety and quality.
Resolian’s specialists have experience handling all types of formulations, including challenging matrices, such as tablets, capsules, creams and ointments, nasal sprays, oligonucleotides, peptides, and other consumer products.
Reach out to us directly to learn more about Resolian’s Analytical Chemistry & Trace Analysis services.
Analytical Chemistry and Trace Analysis Services in Detail
GMP Method Development, Transfer, and Validation: Resolian’s specialist team designs and optimizes novel test methods, routinely utilizing the chromatography and detection options listed below:
- SFC, UPLC, GC and HPLC
- MS, MS/MS, QDa, FLD, FID, ECD, VWD and DAD
CASE STUDY:
Determination of
Highly Polar Impurities in Drug Products
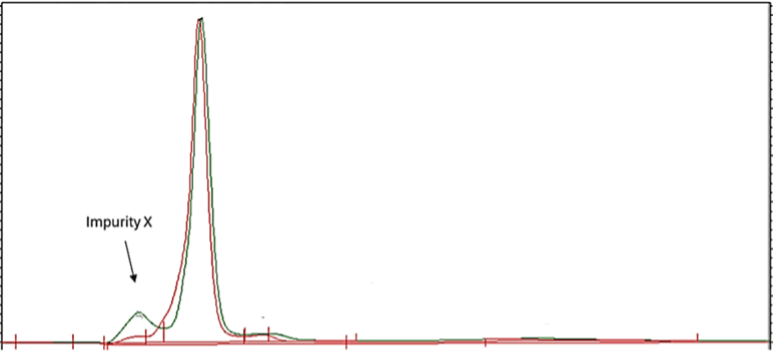
A suitable HPLC-UV method for the determination of Impurity X in Drug Product was required.
- Impurity X has been seen growing on stability in the Drug Product.
- The existing method proved insufficient in resolving Impurity X and any other potential early eluting compounds from the solvent front.
CHALLENGE
-
Impurity X is a very polar compound with poor retention which elutes at the solvent front (refer to Figure 1).
-
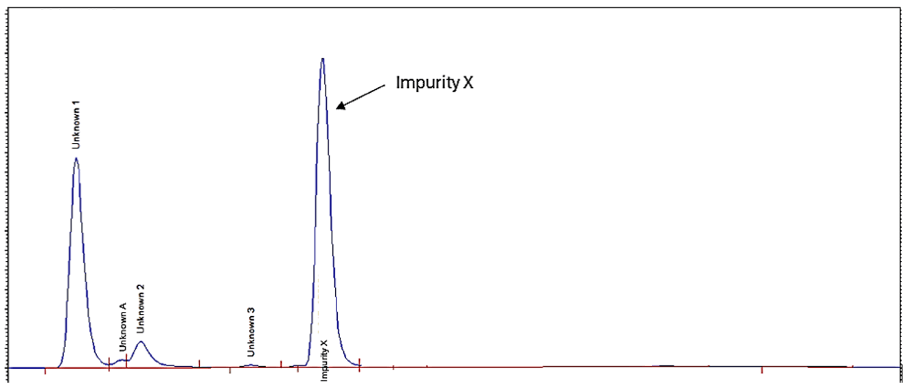
The goal was to develop a method capable of resolving Impurity X and any other potential early eluting compounds from the solvent front and other components (refer to Figure 2).
RESOLIAN APPROACH
- Method Evaluation
- Method Development
- Method Optimization
- Method Validation
SOLUTION
Resolian successfully developed, optimised and validated a GMP method to determine Impurity X using extensive knowledge of method development.
Ready to Solve Your Analytical Chemistry Challenges?
Don’t let inadequate analytical methods compromise your product development timeline or regulatory strategy.
Contact Resolian today to discuss how our analytical sciences team can provide the expertise and solutions you need.
Our Analytical Sciences Resources

News
Resolian and GoBroad Healthcare Group Partnership Streamlines China-Global Clinical Trial Access

Blog, Publications
Contributing to Nitrosamine Science: Resolian’s Role in Beta-Blocker NDSRI Research

Blog, Publications
When AI Meets Experimental Biology: Supporting Breakthrough Peptide Research

Case Studies
Developing and Validating a Simple, Rapid LC-MS/MS Method for Semaglutide in Human Plasma