
Contributing to Nitrosamine Science: Resolian’s Role in Beta-Blocker NDSRI Research
Resolian works alongside drug developers as a trusted guide, delivering nitrosamine testing data to support confident risk decisions.

Resolian works alongside drug developers as a trusted guide, delivering nitrosamine testing data to support confident risk decisions.

Proud to see Resolian’s permeability studies acknowledged in groundbreaking research published in the Journal of Medicinal Chemistry.
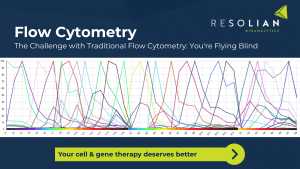
Resolian combines the Cytek Aurora Full Spectrum Profiling platform with 25+ years of flow cytometry expertise to deliver complete immune clarity and confident, regulatory‑ready decisions for cell & gene therapy programs.

Ensure inhalation product safety and compliance with Resolian’s advanced foreign particulate matter characterization and root cause analysis solutions.

Discover how Resolian supports early-career scientists like Jennifer Hawkins through opportunities at the REID Bioanalytical Forum. Learn about LC-MS/MS assay development and career growth in bioanalysis.

Our work, presented at APA 2025, focuses on developing an in vitro CES reaction phenotyping using p-nitrophenyl acetate (p-NPA) as a universal substrate. In this study, we compared different CES substrates, optimized the experimental conditions and developed an LC-MS method for simultaneous detection of p-NPA and its metabolites.

In today’s competitive drug development landscape, contract research organizations (CROs) face mounting pressure to deliver more than just scientific expertise. Success requires a multifaceted approach that combines global reach, consistent delivery, technological innovation, and unwavering client focus.

Ever wondered what happens to your samples once they reach our laboratory?
Take an exclusive behind-the-scenes look at the complete sample journey through our state-of-the-art Fordham facility in Cambridgeshire, UK.
Take a virtual tour of Resolian’s Brisbane bioanalytical laboratory.
Located in the center of Brisbane, Australia, Resolian Bioanalytics is a purpose-built bioanalytical laboratory that can provide comprehensive service to support pre-clinical and clinical studies in Australia and worldwide.

Take a virtual tour of Resolian’s Malvern bioanalytical laboratory – where our expert scientists deliver high-quality data to support nonclinical and clinical trials.

Chief Scientific Officer
Zhiyang Zhao, Ph.D., serves as Chief Scientific Officer (CSO) at Resolian. Dr. Zhao has over 30 years of pharmaceutical industry experience with special focus on drug metabolism and bioanalysis of small and large molecules in drug discovery and development. Dr. Zhao has previously held positions at Pfizer, GlaxoSmithKline, and Amgen. Before joining Resolian in 2015, Dr. Zhao served as Site Director of Preclinical Research at Amgen in Cambridge, Massachusetts, for over a decade.
Currently, Dr. Zhao serves as an Adjunct Professor at the Eshelman School of Pharmacy of the University of North Carolina at Chapel Hill, North Carolina, and as Editor-in-Chief of Drug Metabolism & Bioanalysis Letters, a journal by Bentham Science, which publishes in all areas of drug metabolism and bioanalysis. Dr. Zhao received his Ph.D. degree in Medicinal Chemistry from Virginia Polytechnic and State University (popularly known as Virginia Tech) in Blacksburg, Virginia.

Chief Executive Officer
Patrick Bennett has over 35 years of experience in pharmaceutical analysis and laboratory management. Now Chief Business Officer at Resolian, Patrick’s experience includes the roles of Strategic Marketing Director for Pharma with Thermo Fisher Scientific, LabCorp, and Vice President of Strategy and Development with PPD.
Patrick earned a B.S. degree in Toxicology and a M.S. degree in Pharmacology from the College of Pharmacy and Allied Health at St. John’s University and an M.B.A in International Marketing from the Martin J. Whitman School of Management at Syracuse University.