LC-MS
CASE STUDY
Background
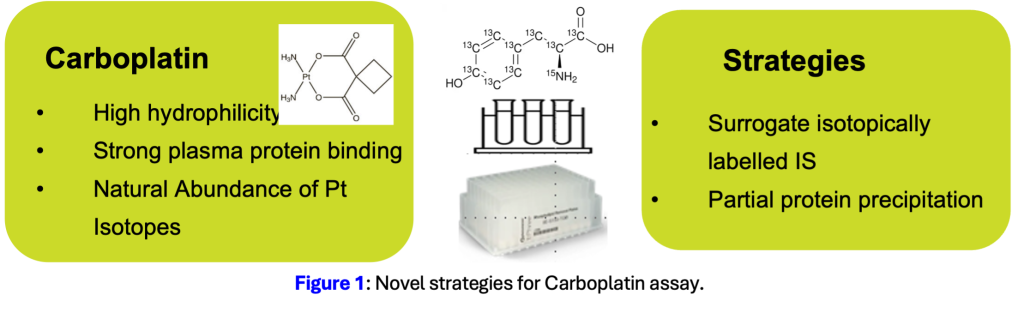
Carboplatin is a platinum-based chemotherapy used in ovarian and non-small cell lung cancer. Getting the dose right matters: accurate measurement of intact drug concentrations is essential for clinical pharmacokinetics and dose optimisation. The problem is that conventional ICP-MS methods measure total platinum, not intact carboplatin, which limits their clinical interpretability. Published LC-MS/MS methods have their own set of challenges: low recovery, isotopic interferences from platinum’s five naturally occurring isotopes, and significant matrix effects that complicate quantitation.
The Challenge
Developing a reliable assay for carboplatin requires navigating three compounding problems simultaneously.
First, standard isotopically labelled internal standards are incompatible: carboplatin-d4 produces overlapping fragment ions due to platinum’s natural isotope cluster, while structural analogues like oxaliplatin fail to correct matrix effects because of poor chromatographic co-elution.
Second, carboplatin’s strong plasma protein binding and high hydrophilicity make it prone to analyte entrapment during sample preparation, suppressing recovery.
Third, its metal sensitivity and polarity make it difficult to retain and separate chromatographically without surface degradation over time.
Our Approach
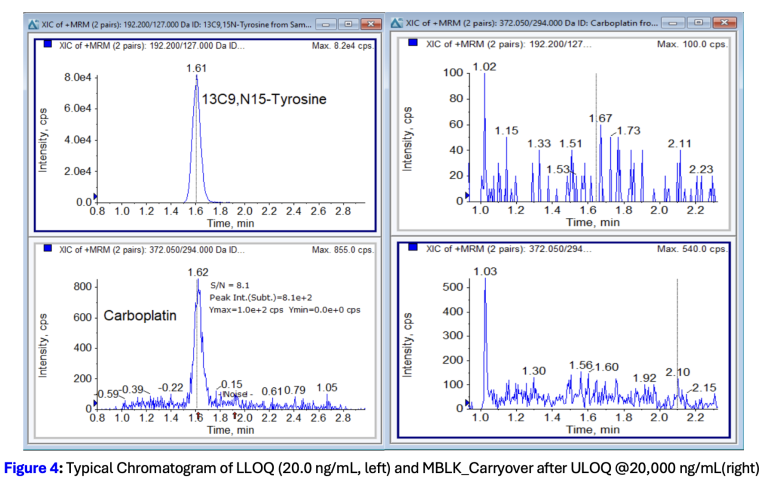
Resolian’s scientists developed a novel LC-MS/MS method that resolves each obstacle through targeted method innovation. After screening 20 internal standard candidates, the team selected 13C9,15N-Tyrosine as a surrogate IS. Unlike carboplatin-d4, it carries no overlapping isotope clusters, and unlike oxaliplatin, it co-elutes with carboplatin to provide reliable matrix-effect correction throughout the run.

For sample preparation, the team combined partial protein precipitation (PPP) with Phree phospholipid removal (PPR). The partial precipitation approach uses a low organic solvent ratio to release carboplatin from protein binding without excessive protein crash, preserving analyte integrity.
The PPR step then removes phospholipids from the extract, producing a consistently clean filtrate and high, stable analyte recovery across batches.
Chromatographic separation was achieved using a Z-HILIC column on MaxPeak hardware, which stabilised retention for this highly polar, metal-sensitive compound and reduced surface interactions that typically degrade peak shape over extended use.
Facing Similar Assay Challenges?
Resolian partners with leading pharma and biotech companies to deliver validated LC-MS/MS methods
for complex small molecules and oncology drugs.
Results
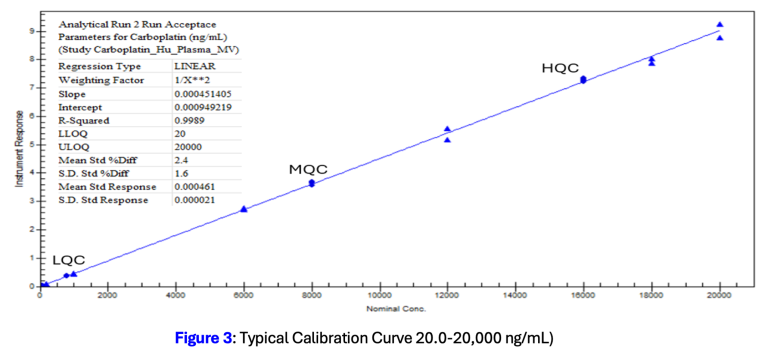
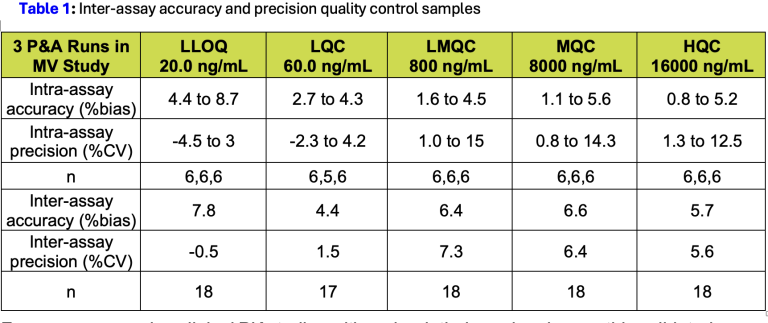
The method was successfully validated to ICH M10 guidance across a linearity range of 20.0 to 20,000 ng/mL, with inter-assay accuracy within 4.4 to 7.8% bias and inter-assay precision below 7.3% CV across all QC levels. Validation confirmed:
- Dilution integrity to 20x
- Selectivity and matrix effect acceptance
- Sample collection stability for 2 hours on wet ice and at room temperature
- Benchtop stability for 19 hours at 4 degrees C
- Freeze-thaw stability across 5 cycles at -70/4 degrees C
- Processed sample stability for 313 hours and reinjection reproducibility for 323 hours at 4 degrees C
The method is validated and ready for deployment in clinical bioanalysis studies.
For sponsors running clinical PK studies with carboplatin-based regimens, this validated assay provides a measurement approach that more accurately reflects the clinically relevant species—intact carboplatin—rather than total platinum.
The combination of a surrogate IS strategy, optimised sample preparation, and robust chromatography delivers the sensitivity and reproducibility needed for regulated bioanalysis, including data that can directly inform dose optimisation decisions for patients.
Ready to Measure What Matters?
Resolian partners with leading pharma and biotech companies to deliver validated LC-MS/MS methods for complex small molecules and oncology drugs.

